Natural mineral allows new method for X-ray imaging
Researchers from the University of Turku, Finland, have discovered a new method of X-ray imaging based on the colouring abilities of the natural mineral hackmanite. The international group of researchers also found out how and why hackmanite changes colour upon exposure to X-rays.
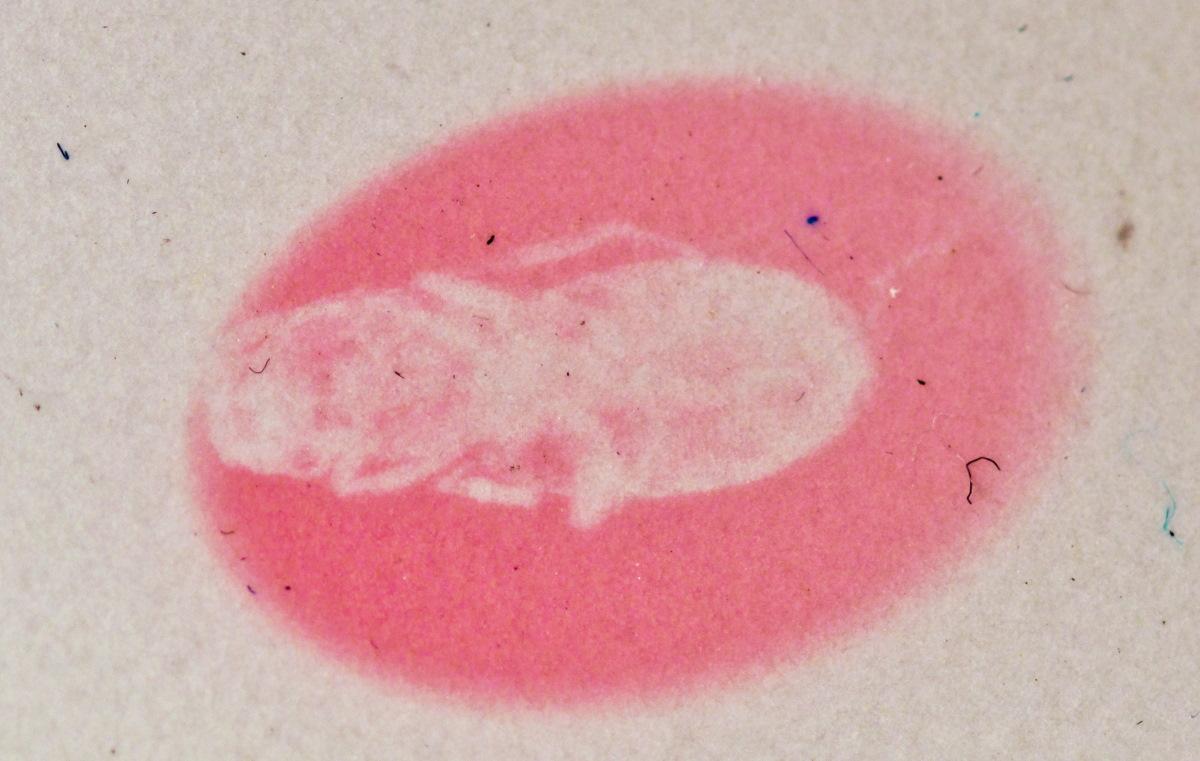
Hackmanite is a natural mineral which glows in the dark. It is also known for its ability to change colour from off-white to pink or violet upon UV exposure. In the recently-published study, the mineral’s colouring abilities were studied under exposure to X-rays.
‘Previous studies had published initial results about the way hackmanite changes colour upon exposure to X-rays, but it was unclear why and how this colouring came to be and whether this feature could be useful somehow,’ says Docent Mika Lastusaari from the University.
‘We were the first to attempt X-ray imaging using a surface made of hackmanite as the image plate, and the results were very exciting.’
The research group for intelligent photonic materials led by Lastusaari conducts pioneering research of materials with light and colour-related properties at the Department of Chemistry of the University of Turku.
The group creates the hackmanites synthetically, which enables them to control the properties of the material and adapt it for different uses by adding or replacing atoms in the basic structure of the material.
Doctoral Candidate Sami Vuori adds, ‘In this research, we studied the behaviour of four types of synthetic hackmanites upon exposure to X-rays in the Karlsruhe synchrotron radiation facility in Germany using a particle accelerator which generates X-rays with tunable energy.
‘All of the samples were positive surprises, and we were able to receive valuable information about how adding different atoms in the basic structure impacts the colouring abilities.
‘We also noticed that the mechanism of colour changing occurs by X-rays exciting the inner shell electrons, unlike with UV radiation, which causes only the loosest electrons of the outer shells to participate in the colouration.’







